Electrostatic force. Coulomb’s Law
The electrostatic force and Coulomb’s law online simulations on this page allow us to visualize how this important force of nature is generated. We will study how its value is calculated from Coulomb’s Law.
This Thematic Unit is part of our Physics collection

STEM OnLine mini dictionary
Coulomb’s Constant
Proportionality constant (k) depending on the medium; in a vacuum, its value is approximately 8.99 · 10⁹ N · m²/C² in the SI.
Coulomb’s Law
Law stating that the force between two charges is proportional to the product of their charges and inversely proportional to the square of the distance: F = k · (q1 · q2) / r².
Electrostatic Attraction
Phenomenon occurring when two charges of opposite signs experience a force tending to pull them together.
Electrostatic Force
Fundamental force of attraction or repulsion acting between electrically charged bodies, described by Coulomb’s law.
Electrostatic Repulsion
Phenomenon occurring when two charges of the same sign experience a force tending to push them apart.
Inverse-Square Law
Principle indicating that the force intensity decreases proportionally to the square of the distance (1/r²) between the charges.
Point Charge
Idealized model of a charged body whose dimensions are negligible compared to the distance of interaction with other charges.
What is electrostatic force
Electrostatic force is a fundamental force in nature that arises from the interaction between electric charges. It is a force of an electrical nature that acts at a distance and can be attractive or repulsive, depending on the properties of the charges involved.
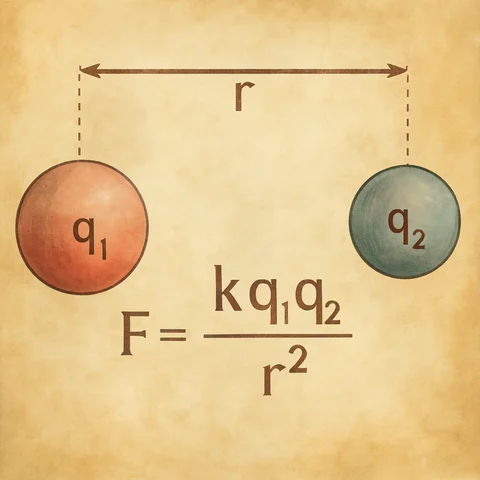
Formula of Coulomb’s law
Electrostatic force is governed by Coulomb’s Law, formulated by the French physicist Charles-Augustin de Coulomb. According to this law, the magnitude of the electrostatic force between two point charges is directly proportional to the product of their charge magnitudes and inversely proportional to the square of the distance separating them. Mathematically, the formula of Coulomb’s law is expressed as:
Electrostatic force = (k x q₁ x q₂) / r².
Where:
Electrostatic force is the magnitude of the force between charges.
k is the Coulomb constant, which depends on the medium in which the charges are located and has a value of approximately 9 × 10^9 N-m²/C² in vacuum.
q₁ and q₂ are the magnitudes of the charges involved.
r is the distance between the charges.
Electrostatic force applications
The electrostatic force is fundamental to many aspects of physics and has numerous applications in everyday life and technology. For example, it is responsible for the interaction between electrons and nuclei in atoms, maintaining the stability of matter. It also determines the structure and properties of chemical compounds.
In the field of electricity and electronics, electrostatic force is essential for the operation of electrical devices, such as capacitors, which store energy in the form of charges separated by a potential difference. It is also the force behind electrostatic phenomena, such as the attraction of charged objects by rubbing or the repulsion between electrically charged balloons.
Electrostatic force is also applied in technologies such as electrodynamics, electricity generation and distribution, and touch screen technology. In addition, it has implications in fields such as astrophysics, where electrostatic interactions are studied in the behavior of stars and galaxies.

STEM OnLine mini dictionary
Coulomb’s Constant
Proportionality constant (k) depending on the medium; in a vacuum, its value is approximately 8.99 · 10⁹ N · m²/C² in the SI.
Coulomb’s Law
Law stating that the force between two charges is proportional to the product of their charges and inversely proportional to the square of the distance: F = k · (q1 · q2) / r².
Electrostatic Attraction
Phenomenon occurring when two charges of opposite signs experience a force tending to pull them together.
Electrostatic Force
Fundamental force of attraction or repulsion acting between electrically charged bodies, described by Coulomb’s law.
Electrostatic Repulsion
Phenomenon occurring when two charges of the same sign experience a force tending to push them apart.
Inverse-Square Law
Principle indicating that the force intensity decreases proportionally to the square of the distance (1/r²) between the charges.
Point Charge
Idealized model of a charged body whose dimensions are negligible compared to the distance of interaction with other charges.
Explore the exciting STEM world with our free, online, simulations and accompanying companion courses! With them you’ll be able to experience and learn hands-on. Take this opportunity to immerse yourself in virtual experiences while advancing your education – awaken your scientific curiosity and discover all that the STEM world has to offer!
Electrostatic force and Coulomb's law simulations
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

André-Marie Ampère
–

Michael Faraday
–
Become a giant


Principles of Modeling, Simulations, and Control for Electric Energy Systems



Principles of Electric Circuits | 电路原理



Electrotechnique I



Electromagnetic Compatibility Essentials



AP® Physics 1: Challenging Concepts



AP® Physics 1 – Part 4: Exam Prep



AP® Physics 1 – Part 2: Rotational Motion



Pre-University Physics

Professional development for Educators


Teaching with Physical Computing: Assessment of Project-Based Learning



Support kids’ projects: Programming with Scratch



Teach teens computing: Impact of technology



Teach teens computing: Machine learning and AI

Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Michael Faraday
–

André-Marie Ampère
–
Become a giant


Principles of Modeling, Simulations, and Control for Electric Energy Systems



Principles of Electric Circuits | 电路原理



Electrotechnique I



Electromagnetic Compatibility Essentials



AP® Physics 2: Challenging Concepts



AP® Physics 1: Challenging Concepts



Pre-University Physics



AP® Physics 1 – Part 1: Linear Motion

Professional development for Educators


AI for Teacher Assistance



Introduction to Online Education & Course Planning



Innovating Instruction: Learning Design in the STEM Classroom



HP Digital Skills for Educators – Google Workspace

Test your knowledge
What is electrostatic force, and how does it explain the interaction between electric charges?
How is Coulomb’s Law formulated, and what does it reveal about the strength of electrostatic interactions?
Why does the force between two charges decrease when the distance increases?
What determines whether the force between two charges is attractive or repulsive?
What role does the constant (k) play in Coulomb’s Law?
You may also be interested
Last simulations
To learn and experience

Take your knowledge to the next level with science kits and hands-on tools that connect theory with experimentation

Van De Graaff generator

Teachers essentials

Notion
Organize your notes, tasks, and projects in one place. Notion combines notes, lists, and calendars in one flexible space.

Canva
Design presentations, diagrams, and infographics with easy-to-use templates and professional results

Genially
Create interactive content, presentations, and educational games in a simple and visual way

Desmos
The most intuitive graphing calculator to explore mathematics and create dynamic classroom activities

Tinkercad
Design in 3D and simulate electronic circuits for free; it’s the perfect tool for simple robotics projects

Your Infinite Library
Enjoy the freedom to explore millions of titles and a curated selection of magazines on any device


Join Prime for students and young adults
The best series. Millions of songs. All included with your subscription.

Science made fun
Learn with the best courses

edX
Online courses from leading universities and companies. Learn practical skills and earn professional certifications

Coursera
Courses from leading universities and companies. Learn practical skills and earn professional certifications

Udemy
Thousands of online courses tailored to your level. Learn at your own pace with expert instructors














