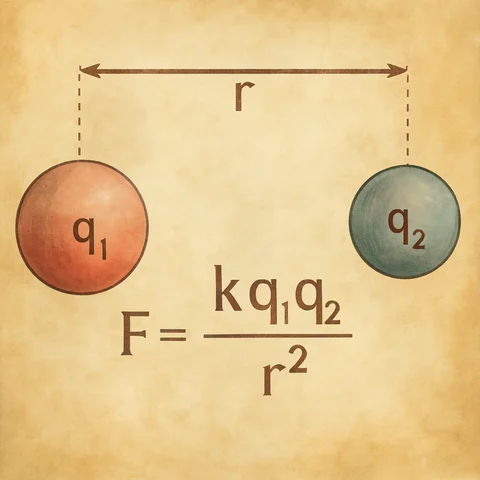Electrostatic force and Coulomb's law simulations
- Force
- Coulomb I
- Coulomb II
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

André-Marie Ampère
1775
–
1836
André-Marie Ampère formulated the theory of electromagnetism, establishing the mathematical foundations linking electricity and magnetism
“Science is the explanation of the complex by the simple”

Michael Faraday
1791
–
1867
Michael Faraday discovered electromagnetic induction, conducted pioneering experiments in optics (Faraday effect), and established fundamental principles of electrochemistry.
“Nothing is too wonderful to be true”
Become a giant
Your path to becoming a giant of knowledge begins with these top free courses

Free mode

Principles of Modeling, Simulations, and Control for Electric Energy Systems


Free mode

Principles of Electric Circuits | 电路原理


Free mode

Electrotechnique I


Free mode

Electromagnetic Compatibility Essentials


Free mode

Pre-University Physics


Free mode

Circuits for Beginners


Free mode

AP® Physics 1: Challenging Concepts


Free mode

AP® Physics 1 – Part 4: Exam Prep

Professional development for Educators
Your path to becoming a giant of knowledge begins with these top free courses

Free mode

Teach teens computing: How computers work


Free mode

Get started with teaching computing


Free mode

Support kids’ projects: Web development


Free mode

Teaching with Physical Computing: Soft skills, teamwork and the wider curriculum