Simulations and 3d molecules of chemical salts
- Solubility
- Sodium
- Copper
- Thallium
Salts and solubility
Add different salts to water, and then watch them dissolve and achieve dynamic equilibrium with the solid precipitate. Compare the number of ions in the highly soluble NaCl solution with other poorly soluble salts. Relate the charges on the ions to the number of ions in the formula of a salt. Calculate the Ksp values.
Sodium Chloride
Sodium chloride, common salt or table salt, referred to in its mineral form as halite, is a chemical compound with the formula NaCl. Sodium chloride is one of the salts responsible for the salinity of the ocean and the extracellular fluid of many organisms. It is also the component of common salt, used as a seasoning and food preservative.
Sodium Chloride
NaCl
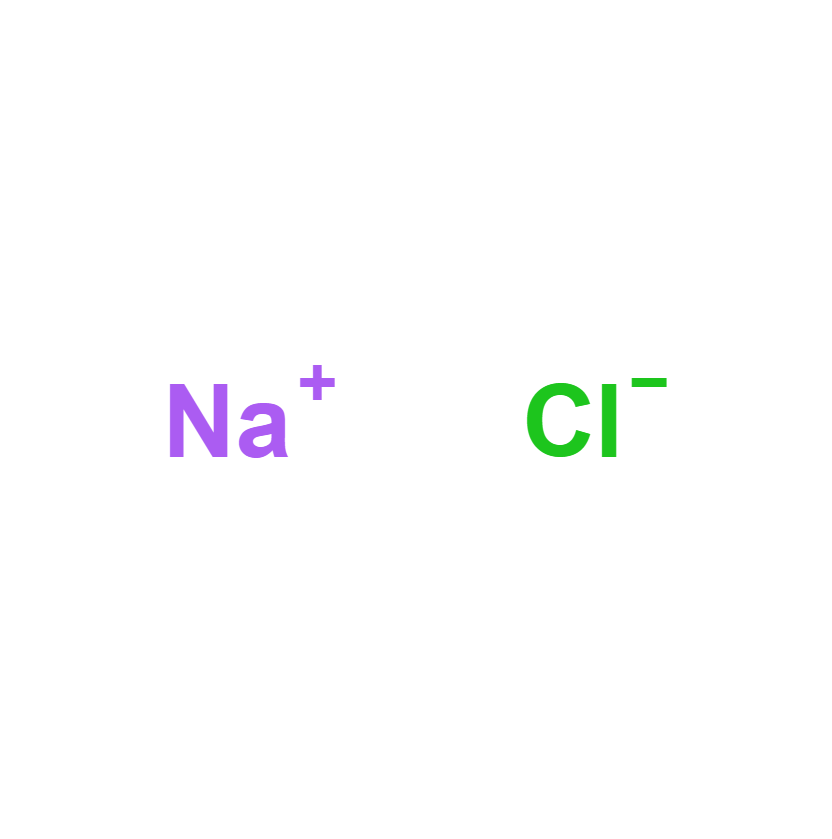
 Single bond
Single bond
Double bond
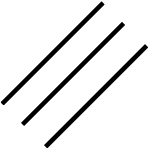 Triple bond
Triple bond
Wedge bond

Hash bond
Copper(I) Iodide
Copper(I) iodide is the inorganic compound with the formula CuI. It is also known as cuprous iodide. It is useful in a variety of applications ranging from organic synthesis to cloud seeding.
Copper(I) Iodide
CuI
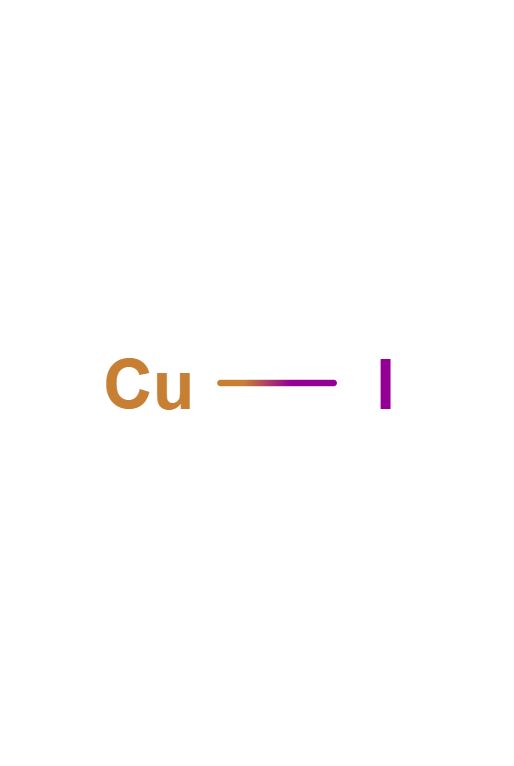
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

John Dalton
1766
–
1844
John Dalton formulated the first atomic theory, explaining the composition of matter and the law of multiple proportions in chemical compounds.
“Science is the foundation of truth”

William Henry
1774
–
1836
William Henry formulated Henry’s Law, describing how the amount of gas dissolved in a liquid is proportional to its partial pressure
“Chemistry reveals the hidden secrets in the invisible”
Become a giant
Your path to becoming a giant of knowledge begins with these top free courses

Free mode

Introduction to Solid State Chemistry


Free mode

Preparing for CLEP Chemistry: Part 1


Free mode

Pre-University Chemistry


Free mode

Big Bang and the Origin of Chemical Elements

Professional development for Educators
Your path to becoming a giant of knowledge begins with these top free courses

Free mode

Teach kids computing: Programming


Free mode

Teach teens computing: Object-oriented Programming in Python


Free mode

An Introduction to Evidence-Based Undergraduate STEM Teaching


Free mode

Support kids’ projects: Web development