Water chemistry. Molecule and properties
The online water chemistry simulations on this page will help you to learn about this very important compound of nature. We will examine what the water molecule is like, what are some of its main chemical properties and we will see in a practical way the phase changes of water changes state, how it is distilled and what is the electrolysis of water..

This Thematic Unit is part of our Chemistry collection

STEM OnLine mini dictionary
Amphoterism
Chemical ability of water to act as an acid or a base depending on the substance it reacts with.
Hydrogen Bond
Especially strong dipole-dipole interaction between the hydrogen atom of one water molecule and the oxygen of another.
Polar Molecule
Molecule that presents an asymmetric distribution of its electrical charges, creating a positive and a negative pole.
Surface Tension
Attraction force between the molecules on the water surface that creates a resistant layer.
Universal Solvent
Ability of water to dissolve a wide variety of substances due to its high dielectric constant and polarity.
Water
Inorganic compound formed by two hydrogen atoms and one oxygen atom (H₂O), essential for all known life forms.
What is water chemistry
Water is an odorless, colorless and tasteless substance. It is a chemical compound essential for life, with unique and diverse properties that make it crucial for a wide range of processes and applications.
Water chemistry is the branch of chemistry dedicated to the study of the properties, structure and behavior of this essential compound for life. This field analyzes from its molecular composition to its interaction with other substances, covering processes such as dissolution, ionization and changes of state. It also explores its role in biological, industrial and environmental systems, being crucial to understand phenomena such as the formation of solutions, chemical reactions in aqueous media and its function as a thermal regulator on the planet.
Water molecule
The water molecule is composed of two hydrogen atoms and one oxygen atom, which is represented by the chemical formula H2O. It is a polar compound, which means that the electrical charge is distributed unequally between the hydrogen and oxygen atoms.
Chemical properties of water
The chemical properties of water are extraordinary and make it a unique and essential compound for life on Earth. Its ability to dissolve various substances, act as a thermal regulator and participate in chemical reactions makes it indispensable in biological, industrial and environmental processes. These characteristics arise from its molecular structure and the bonds formed between its molecules, which generate behaviors that not only guarantee the existence of living beings, but also enable countless scientific applications and discoveries.
Water as a universal solvent
Water is a universal solvent, that is, it has the ability to dissolve a wide variety of substances, including salts, sugars, gases and other organic and inorganic compounds. This property is due to the ability of the opposite poles of water molecules to attract and surround the ions and molecules of other substances, separating and dissolving them in solution.
Water as an amphoteric compound
Water is also an amphoteric compound and therefore can act as either an acid or a base. This is due to the ability of water molecules to ionize, releasing hydrogen ions (H+) and hydroxide ions (OH-) into solution. When an acid is dissolved in water, an excess of H+ ions is produced, while when a base is dissolved, an excess of OH- ions is produced.
Heat capacity of water
Water also has a high heat capacity so a large amount of energy is required to raise its temperature. The reason lies in the bonding forces between water molecules, which absorb energy on heating and release it on cooling.
In summary, the online water chemistry simulations on this page are an excellent tool to learn more about the physical and chemical properties and characteristics of water and the water molecule.

STEM OnLine mini dictionary
Amphoterism
Chemical ability of water to act as an acid or a base depending on the substance it reacts with.
Hydrogen Bond
Especially strong dipole-dipole interaction between the hydrogen atom of one water molecule and the oxygen of another.
Polar Molecule
Molecule that presents an asymmetric distribution of its electrical charges, creating a positive and a negative pole.
Surface Tension
Attraction force between the molecules on the water surface that creates a resistant layer.
Universal Solvent
Ability of water to dissolve a wide variety of substances due to its high dielectric constant and polarity.
Water
Inorganic compound formed by two hydrogen atoms and one oxygen atom (H₂O), essential for all known life forms.
Explore the exciting STEM world with our free, online, simulations and accompanying companion courses! With them you’ll be able to experience and learn hands-on. Take this opportunity to immerse yourself in virtual experiences while advancing your education – awaken your scientific curiosity and discover all that the STEM world has to offer!
Water Chemistry Simulations
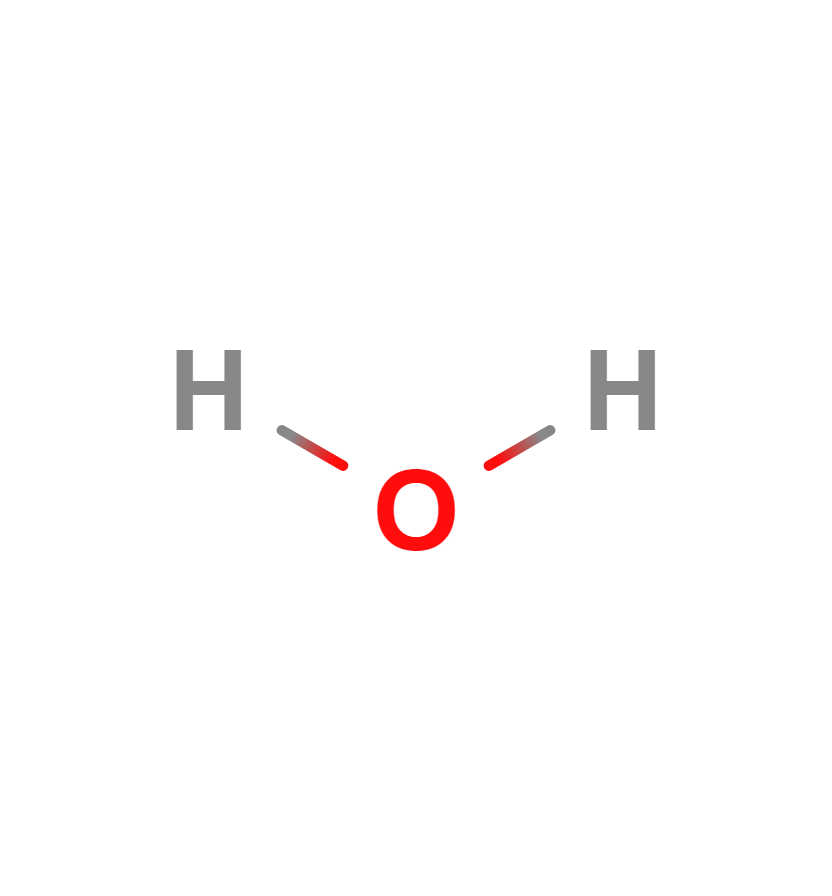
Water molecule
In the first of our online water chemistry simulations we can see that water molecule is composed of two hydrogen atoms and one oxygen atom joined by a covalent bond. That is, the two hydrogen atoms and the oxygen atom are bonded together by sharing electrons. Its formula is H2O.
Water
 Single bond
Single bond
Double bond
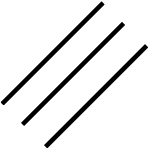 Triple bond
Triple bond
Wedge bond

Hash bond
Electrolysis of water
The electrolysis of water is the decomposition of water (H2O) into the gases oxygen (O2) and hydrogen (H2) by means of a direct electric current. To reduce the resistance to the passage of current through water, it is usually acidified by adding a small amount of sulfuric acid or by adding a strong electrolyte such as sodium hydroxide, NaOH.
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Marie Curie
–

Antoine-Laurent de Lavoisier
–
Become a giant


Introduction to Computational Materials Design



From Atoms to Materials: Predictive Theory and Simulations



Digital Biomaterials



Cement Chemistry and Sustainable Cementitious Materials



Pre-University Chemistry



Preparing for CLEP Chemistry: Part 1



Big Bang and the Origin of Chemical Elements

Professional development for Educators


Interdisciplinary Teaching with Museum Objects



Teaching with Physical Computing: Introduction to Project-Based Learning



Teaching With Technology and Inquiry: An Open Course For Teachers



Teach teens computing: Computer networks

Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Marie Curie
–

John Dalton
–
Become a giant


Introduction to Computational Materials Design



From Atoms to Materials: Predictive Theory and Simulations



Digital Biomaterials



Cement Chemistry and Sustainable Cementitious Materials



Pre-University Chemistry



Preparing for CLEP Chemistry: Part 1



Big Bang and the Origin of Chemical Elements

Professional development for Educators


Teach teens computing: Developing your programming pedagogy



Teaching Computational Thinking



Teach computing: Support SEND learners with computing



Teach teens computing: Functions and algorithms, searching and sorting in Python

Test your knowledge
What does the chemistry of water study, and why is this compound so important in natural and industrial systems?
What is the molecular structure of water, and what implications does its polarity have for its chemical behavior?
Why do people call water a “universal solvent”? It sounds like it can dissolve everything.
What does it mean that water is amphoteric? I get confused with the idea that it can be an acid and a base at the same time.
Why is it so hard to heat water? Sometimes it feels like it takes forever to boil.
You may also be interested
Last simulations
To learn and experience

Take your knowledge to the next level with science kits and hands-on tools that connect theory with experimentation

Electrolysis equipment

Essential for teachers and students

Notion
Organize your notes, tasks, and projects in one place. Notion combines notes, lists, and calendars in one flexible space.

Canva
Design presentations, diagrams, and infographics with easy-to-use templates and professional results

Genially
Create interactive content, presentations, and educational games in a simple and visual way

Desmos
The most intuitive graphing calculator to explore mathematics and create dynamic classroom activities

Tinkercad
Design in 3D and simulate electronic circuits for free; it’s the perfect tool for simple robotics projects

Your Infinite Library
Enjoy the freedom to explore millions of titles and a curated selection of magazines on any device


Join Prime for students and young adults
The best series. Millions of songs. All included with your subscription.

Science made fun
Learn with the best courses

edX
Online courses from leading universities and companies. Learn practical skills and earn professional certifications

Coursera
Courses from leading universities and companies. Learn practical skills and earn professional certifications

Udemy
Thousands of online courses tailored to your level. Learn at your own pace with expert instructors












