Hydrogen. Atom, molecule and properties
The online hydrogen simulations on this page will help you learn about his very important element of nature. We will discover what the atomic structure of hydrogen is like and we will see the atomic model of hydrogen. We will also learn what the hydrogen molecule looks like and some of its most important properties and characteristics

This Thematic Unit is part of our Chemistry collection

STEM OnLine mini dictionary
Deuterium
Stable isotope of hydrogen containing one proton and one neutron in its nucleus.
Diatomic Gas
Molecule formed by two atoms of the same element, as occurs with elemental hydrogen in nature.
Hydride
Chemical compound formed by the combination of hydrogen with any other element, whether metallic or non-metallic.
Hydrogen
Chemical element with atomic number 1, the lightest in the periodic table, which under normal conditions occurs as a diatomic gas (H₂).
Isotope
Atoms of the same element with the same number of protons but a different number of neutrons. Hydrogen has three: protium, deuterium, and tritium.
Tritium
Radioactive isotope of hydrogen whose nucleus consists of one proton and two neutrons.
What is hydrogen
Hydrogen is a chemical element with an atomic number of 1, which means that it has a single proton in its nucleus and a single electron in its valence shell. In its molecular form, hydrogen is in the form of H2, which consists of two hydrogen atoms joined by a covalent bond. It is a diatomic molecule, two atoms of the same element bonding together to form a molecule.
Hydrogen atom. Atomic model of hydrogen
The hydrogen atom, the simplest atom in the universe, has been essential to the development of atomic theory. With a nucleus consisting of only one proton and one electron orbiting around it, the atomic model of hydrogen has served as the basis for understanding the structure of atoms in general. From Bohr’s model, which describes the electron moving in discrete energy levels, to modern models based on quantum mechanics, the study of hydrogen has illuminated the fundamental principles of chemistry and physics. Experiments such as light scattering on the hydrogen atom have been crucial in validating these theories, allowing us to explore the most intriguing properties of this essential element.
Hydrogen molecule
The hydrogen molecule, represented as H2, is one of the simplest and most essential molecules in nature. It is made up of two hydrogen atoms that share a pair of electrons, creating a covalent bond that holds them together. This bonding allows them to achieve a more stable electronic configuration, making molecular hydrogen a key element in countless chemical and biological processes. In addition, its diatomic structure and unique properties make it a fundamental component in the study of the behavior of gases and in various technological applications.
Properties of hydrogen
Hydrogen is the lightest and most abundant element in the universe. Under standard conditions of temperature and pressure, it is a colorless, odorless and highly flammable gas. It is highly reactive and can form bonds with many other elements, including carbon, nitrogen, oxygen and halogens.
Chemical relevance
Hydrogen is an important element in organic chemistry, as it is a key component of hydrocarbon molecules, which form the basis of fossil fuels. It is part of the water molecule (H2O) and other chemical compounds essential to life. Its ability to form compounds with different elements makes it a key intermediate in many industrial chemical reactions, such as the production of ammonia by the Haber-Bosch process.
Physical properties
In terms of physical properties, hydrogen has a very low boiling point (-252.87°C) and an even lower melting point (-259.16°C). Although it is a gas at normal temperature and pressure, it can be liquefied at extremely low temperatures and stored in liquid form for use in industrial and transportation applications. It should be noted that hydrogen has a very low density, which makes it difficult to store and transport efficiently, but at the same time makes it ideal for applications such as research balloons and space rockets.
Thermal capacity and conductivity
Hydrogen is an excellent conductor of heat and electricity and has a high heat capacity. This makes it an attractive energy medium in advanced heat transfer systems. In addition, its low viscosity and high diffusion make it ideal for applications in fuel cell technology.
Hydrogen isotopes
A unique property of hydrogen is the existence of its three isotopes: protium, deuterium and tritium. Protium is the most common isotope, while deuterium is used in applications such as nuclear reactors and fusion research. Tritium, although radioactive, plays a key role in nuclear fusion experiments and in the illumination of night vision devices.
Taken together, all these properties make hydrogen both scientifically and technologically exciting, with applications ranging from basic research to the development of sustainable energy solutions.

STEM OnLine mini dictionary
Deuterium
Stable isotope of hydrogen containing one proton and one neutron in its nucleus.
Diatomic Gas
Molecule formed by two atoms of the same element, as occurs with elemental hydrogen in nature.
Hydride
Chemical compound formed by the combination of hydrogen with any other element, whether metallic or non-metallic.
Hydrogen
Chemical element with atomic number 1, the lightest in the periodic table, which under normal conditions occurs as a diatomic gas (H₂).
Isotope
Atoms of the same element with the same number of protons but a different number of neutrons. Hydrogen has three: protium, deuterium, and tritium.
Tritium
Radioactive isotope of hydrogen whose nucleus consists of one proton and two neutrons.
Explore the exciting STEM world with our free, online, simulations and accompanying companion courses! With them you’ll be able to experience and learn hands-on. Take this opportunity to immerse yourself in virtual experiences while advancing your education – awaken your scientific curiosity and discover all that the STEM world has to offer!
Hydrogen molecule
Hydrogen is the chemical element of atomic number 1, represented by the symbol H. It usually occurs in its molecular form. , The hydrogen molecule is made up of two hydrogen atoms, which gives rise to the diatomic gas H2 under normal conditions. This gas is flammable, colorless, odorless, non-metallic and insoluble in water.
Hydrogen
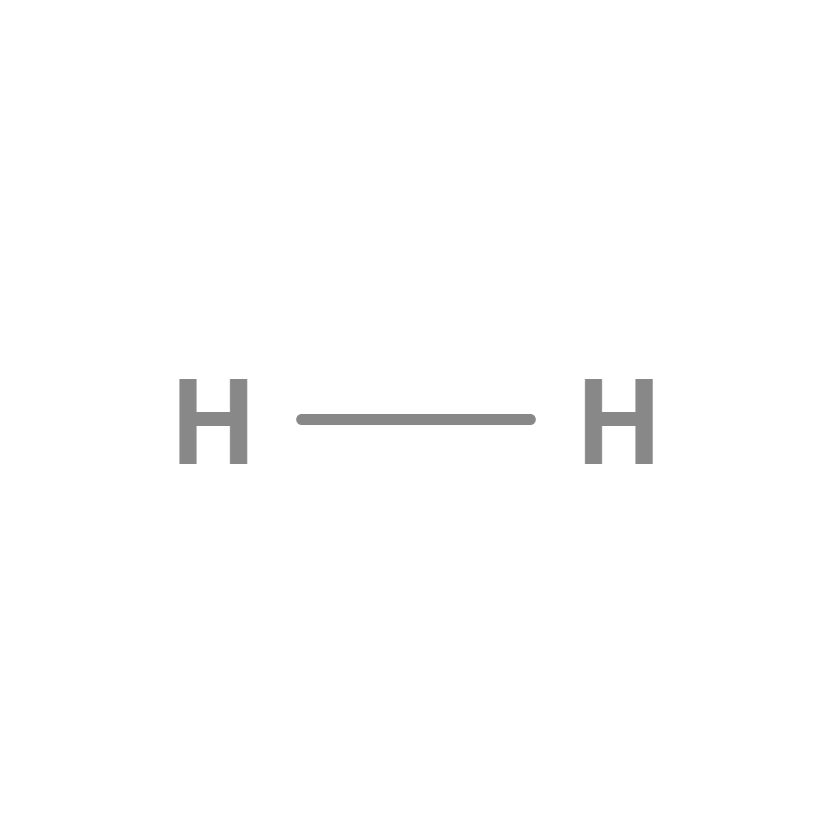
 Single bond
Single bond
Double bond
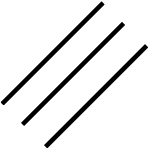 Triple bond
Triple bond
Wedge bond

Hash bond
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

John Dalton
–

Fritz Haber
–
Become a giant


Introduction to Computational Materials Design



From Atoms to Materials: Predictive Theory and Simulations



Digital Biomaterials



Cement Chemistry and Sustainable Cementitious Materials



Preparing for CLEP Chemistry: Part 1



Pre-University Chemistry



Big Bang and the Origin of Chemical Elements

Professional development for Educators


Teach teens computing: Databases and SQL



AI for Teacher Assistance



Teach teens computing: Object-oriented Programming in Python



STEM Outside

Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Marie Curie
–

Antoine-Laurent de Lavoisier
–
Become a giant


Introduction to Computational Materials Design



From Atoms to Materials: Predictive Theory and Simulations



Digital Biomaterials



Cement Chemistry and Sustainable Cementitious Materials



Preparing for CLEP Chemistry: Part 1



Big Bang and the Origin of Chemical Elements



Pre-University Chemistry

Professional development for Educators


AI for Teacher Assistance



Advancing Learning Through Evidence-Based STEM Teaching



Higher education assessing in the age of AI



Teach kids computing: Developing your programming pedagogy

Test your knowledge
What is hydrogen, and how is it characterized at the atomic and molecular level?
How has the hydrogen atom contributed to the development of atomic models?
Why is the hydrogen molecule H₂ so important?
What are the most important properties of hydrogen?
What is hydrogen used for, and why is it so relevant today?
You may also be interested
Last simulations
To learn and experience

Take your knowledge to the next level with science kits and hands-on tools that connect theory with experimentation

Hydrogen fuel cell

Essential for teachers and students

Notion
Organize your notes, tasks, and projects in one place. Notion combines notes, lists, and calendars in one flexible space.

Canva
Design presentations, diagrams, and infographics with easy-to-use templates and professional results

Genially
Create interactive content, presentations, and educational games in a simple and visual way

Desmos
The most intuitive graphing calculator to explore mathematics and create dynamic classroom activities

Tinkercad
Design in 3D and simulate electronic circuits for free; it’s the perfect tool for simple robotics projects

Your Infinite Library
Enjoy the freedom to explore millions of titles and a curated selection of magazines on any device


Join Prime for students and young adults
The best series. Millions of songs. All included with your subscription.

Science made fun
Learn with the best courses

edX
Online courses from leading universities and companies. Learn practical skills and earn professional certifications

Coursera
Courses from leading universities and companies. Learn practical skills and earn professional certifications

Udemy
Thousands of online courses tailored to your level. Learn at your own pace with expert instructors













