Greenhouse effect. Mechanism, model and gases
The online greenhouse effect simulations on this page serve as a model of how the greenhouse effect works and show the mechanism of the greenhouse effect. They also show some of the main greenhouse gases.

This Thematic Unit is part of our Earth Sciences collection

STEM OnLine mini dictionary
Carbon Dioxide
Main greenhouse gas generated by burning fossil fuels and respiration.
Global Warming
Increase in the Earth’s average temperature due to the rise in greenhouse gases.
Greenhouse Effect
Natural process where certain gases trap solar heat to maintain a habitable temperature.
Greenhouse Gases
Gaseous components that trap thermal radiation and regulate the planet’s temperature.
Infrared Radiation
Heat emitted by the Earth’s surface that is absorbed by greenhouse gases.
Methane
Potent greenhouse gas produced by organic decomposition and agricultural activities.
Radiative Forcing
Change in the atmosphere’s net energy flow caused by climate factors.
Solar Radiation
Energy in the form of light and heat emitted by the Sun that reaches the Earth.
Thermal Balance
Equilibrium between incoming solar energy and heat energy leaving for space.
Water Vapor
Water in a gaseous state that mixes with the air; it is the main driver of cloud formation and meteorological phenomena.
What is the greenhouse effect
The greenhouse effect is a natural process in which certain gases in the Earth’s atmosphere retain part of the infrared radiation emitted by the Earth after receiving solar energy. This causes the atmosphere to warm, allowing the surface temperature to become suitable for life.
The scientific basis for the greenhouse effect has been supported by multiple lines of evidence, including direct measurements of gas concentrations in the atmosphere, studies of infrared radiation, and models of the greenhouse effect that simulate the behavior of the Earth in response to changes in greenhouse gas levels.
How the greenhouse effect works. Mechanism of the greenhouse effect
Basic mechanism of the greenhouse effect is as follows. Solar radiation reaching the Earth consists mainly of short-wave radiation, such as visible light and ultraviolet (UV) rays. Some of this radiation is reflected back into space by the Earth’s surface and clouds, while some is absorbed by the atmosphere, the oceans and the Earth’s surface.
The Earth’s surface, once heated by solar radiation, emits long-wave radiation, also known as infrared (IR) radiation. Some gases present in the atmosphere have the capacity to absorb and re-emit part of this infrared radiation.
Main greenhouse gases
The main greenhouse gases are water vapor (H2O), carbon dioxide (CO2), methane (CH4) or nitrous oxide (N2O) have the ability to absorb and re-emit part of this infrared radiation. These gases are known as greenhouse gases because of their ability to trap and retain heat in the atmosphere. When infrared radiation is absorbed by greenhouse gases, the molecules become energized and subsequently emit radiation back in all directions, including towards the earth’s surface. This results in a net increase in thermal energy on Earth, leading to an increase in atmospheric and surface temperature.

STEM OnLine mini dictionary
Carbon Dioxide
Main greenhouse gas generated by burning fossil fuels and respiration.
Global Warming
Increase in the Earth’s average temperature due to the rise in greenhouse gases.
Greenhouse Effect
Natural process where certain gases trap solar heat to maintain a habitable temperature.
Greenhouse Gases
Gaseous components that trap thermal radiation and regulate the planet’s temperature.
Infrared Radiation
Heat emitted by the Earth’s surface that is absorbed by greenhouse gases.
Methane
Potent greenhouse gas produced by organic decomposition and agricultural activities.
Radiative Forcing
Change in the atmosphere’s net energy flow caused by climate factors.
Solar Radiation
Energy in the form of light and heat emitted by the Sun that reaches the Earth.
Thermal Balance
Equilibrium between incoming solar energy and heat energy leaving for space.
Water Vapor
Water in a gaseous state that mixes with the air; it is the main driver of cloud formation and meteorological phenomena.
Explore the exciting STEM world with our free, online, simulations and accompanying companion courses! With them you’ll be able to experience and learn hands-on. Take this opportunity to immerse yourself in virtual experiences while advancing your education – awaken your scientific curiosity and discover all that the STEM world has to offer!
Greenhouse effect simulations
Greenhouse effect model
This simulation is a simple but very instructive model of the mechanism of the greenhouse effect. How do greenhouse gases affect the climate? Explore the atmosphere during the ice age and today. What happens when clouds are added? Change the concentration of greenhouse gases and see how the temperature changes. Then compare the effect of crystals. Zoom in and see how light interacts with molecules Do all gases in the atmosphere contribute to the greenhouse effect?
Water vapor
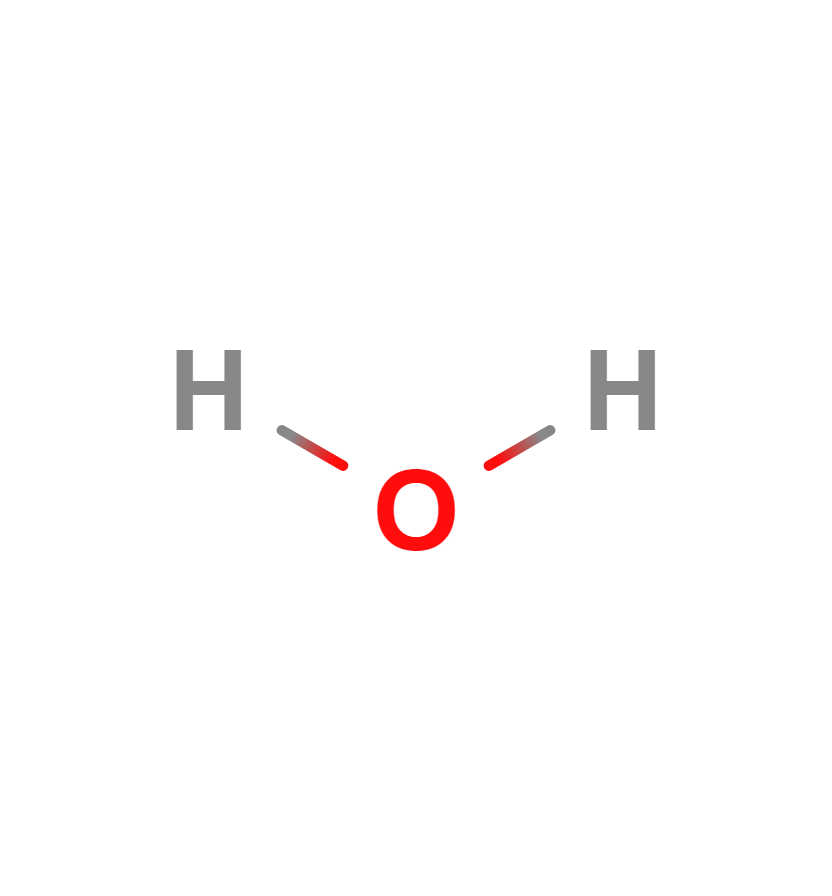
Water vapor is an important greenhouse gas. The water molecule is composed of two hydrogen atoms and one oxygen atom bonded together by a covalent bond. That is, the two hydrogen atoms and the oxygen atom are bonded together by sharing electrons. Its formula is H2O.
Water
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
Carbon dioxide
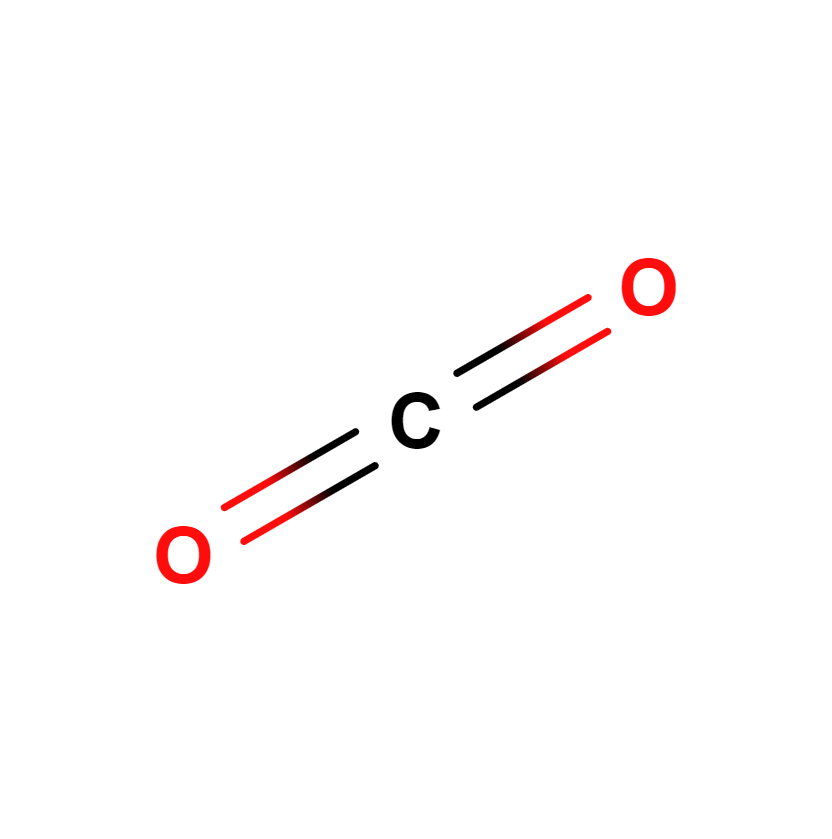
Carbon dioxide, whose chemical formula is CO2, is a compound of carbon and oxygen that exists as a colorless gas at standard temperature and pressure conditions. Prior to the 2005 IUPAC standards, it was also known as carbonic anhydride.
Carbon dioxide
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
Methane
Methane is the simplest alkane hydrocarbon, whose chemical formula is CH4. Each of the hydrogen atoms is bonded to carbon by a covalent bond. It is a non-polar substance that occurs as a gas at ordinary temperatures and pressures. It is colorless, odorless and insoluble in water.
Methane
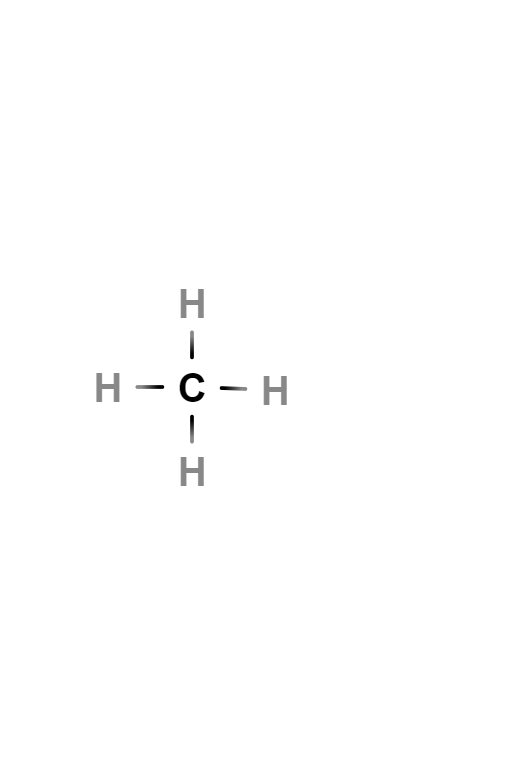
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
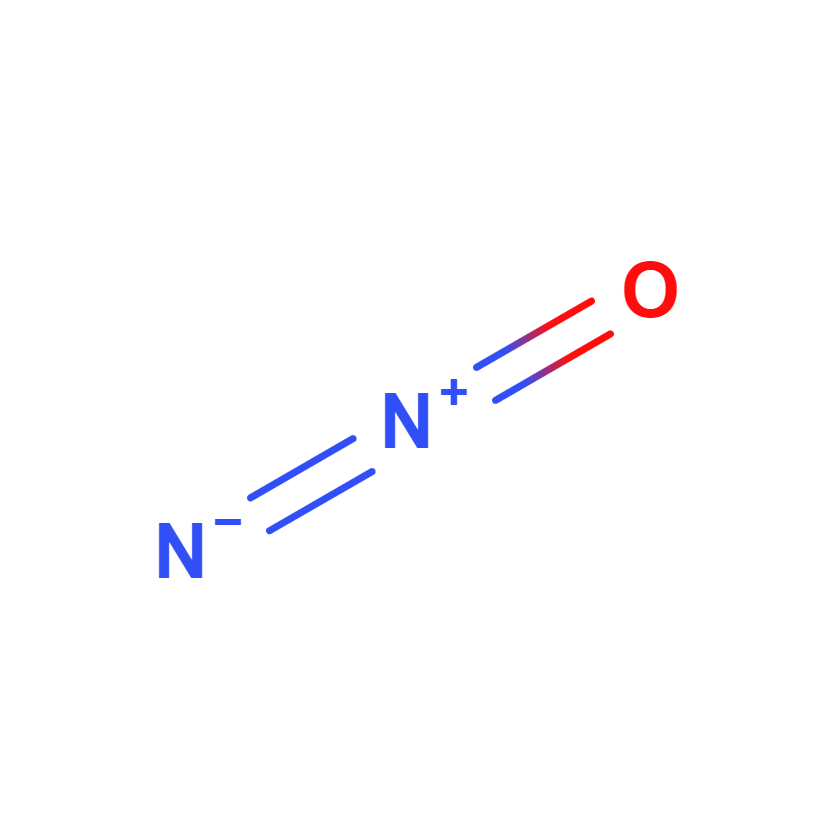
Nitrous oxide
Nitrous oxide, whose chemical formula is N2O, also known as laughing gas, is a colorless gas with a sweet, slightly toxic odor and anesthetic effect. It is one of the most important greenhouse gases and is a contributor to stratospheric ozone depletion.
Nitrous oxide
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Vilhelm Friman Koren Bjerknes
–

Alfred Lothar Wegener
–
Become a giant


Modeling Climate Change



Introduction to Water and Climate



Global Warming Science



Climate Change: The Science and Global Impact



Sensing Planet Earth – Water and Ice



The History of Ancient Environments, Climate, and Life



Introduction to Deep Earth Science



Our Global Ocean – An Introduction Course

Professional development for Educators


Support kids’ projects: Programming with Scratch



Teaching and Learning in the Era of AI



Get started with your Raspberry Pi computer



Teach teens computing: Programming in Python

Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Vilhelm Friman Koren Bjerknes
–

Alexander von Humboldt
–
Become a giant


Modeling Climate Change



Introduction to Water and Climate



Global Warming Science



Climate Change: The Science and Global Impact



Introduction to Deep Earth Science



Sensing Planet Earth – Water and Ice



The History of Ancient Environments, Climate, and Life



Sensing Planet Earth – From Core to Outer Space

Professional development for Educators


Teach teens computing: Impact of technology



AI for Teacher Assistance



Support kids’ projects: Programming with Scratch



Teaching Computational Thinking

Test your knowledge
What is the greenhouse effect?
How does the greenhouse effect work in the atmosphere?
Why is the greenhouse effect important for life on Earth?
Which gases maintain the natural greenhouse effect?
What would happen if the greenhouse effect did not exist?
You may also be interested
Last simulations
To learn and experience

Take your knowledge to the next level with science kits and hands-on tools that connect theory with experimentation
Air quality monitor

Teachers essentials

Notion
Organize your notes, tasks, and projects in one place. Notion combines notes, lists, and calendars in one flexible space.

Canva
Design presentations, diagrams, and infographics with easy-to-use templates and professional results

Genially
Create interactive content, presentations, and educational games in a simple and visual way

Desmos
The most intuitive graphing calculator to explore mathematics and create dynamic classroom activities

Tinkercad
Design in 3D and simulate electronic circuits for free; it’s the perfect tool for simple robotics projects

Your Infinite Library
Enjoy the freedom to explore millions of titles and a curated selection of magazines on any device


Join Prime for students and young adults
The best series. Millions of songs. All included with your subscription.

Science made fun
Learn with the best courses

edX
Online courses from leading universities and companies. Learn practical skills and earn professional certifications

Coursera
Courses from leading universities and companies. Learn practical skills and earn professional certifications

Udemy
Thousands of online courses tailored to your level. Learn at your own pace with expert instructors













