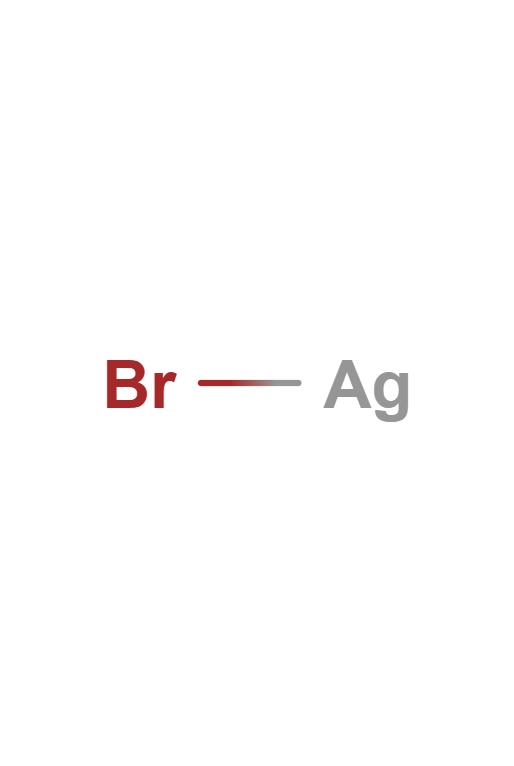3d molecules of chemical salts
- Silver
- Mercury
- Strontium
- Silver II
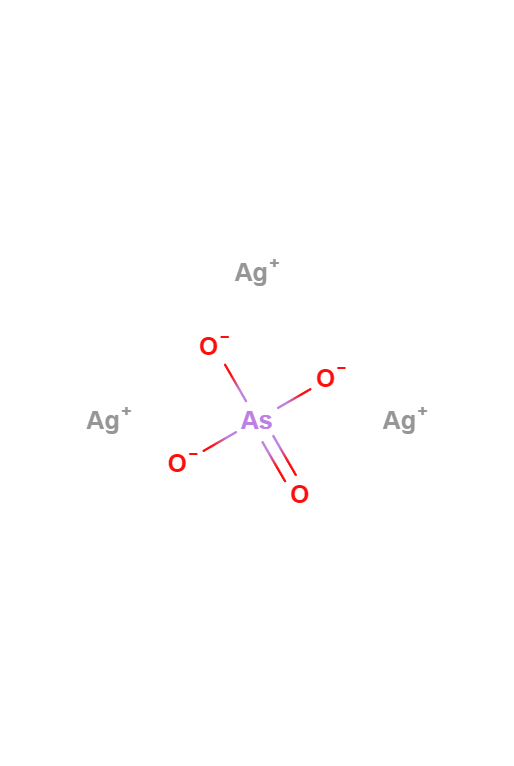
Silver Arsenate
Silver Arsenate is an inorganic compound with the formula Ag3AsO4. It has been used in qualitative analysis to distinguish between phosphate and arsenate solutions.
Silver arsenate
Ag3AsO4
 Single bond
Single bond
Double bond
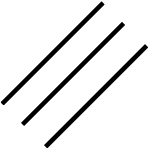 Triple bond
Triple bond
Wedge bond

Hash bond
Mercury (I) Bromide
Mercury (I) Bromide is a chemical compound with the chemical formula Hg2Br2. It changes color from white to yellow when heated and fluoresces salmon-colored when exposed to ultraviolet light. It has applications in acoustic-optical devices.
Mercury(I) Bromide
Hg2Br2
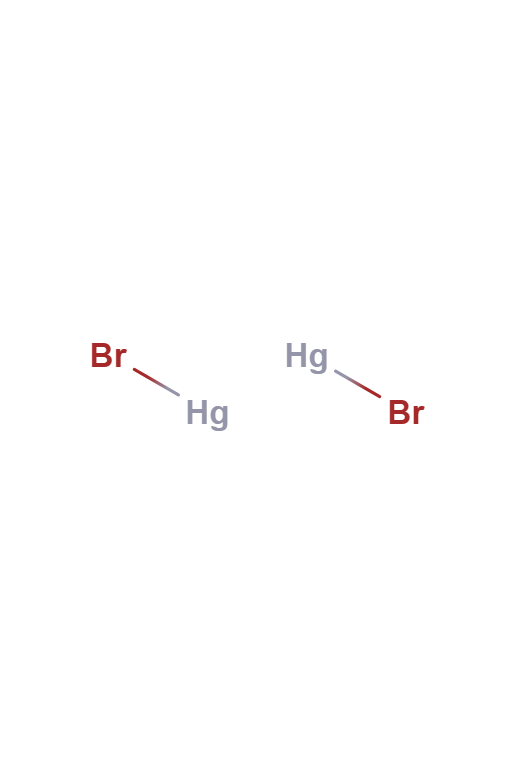
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
Strontium Phosphate
Strontium Phosphate is a chemical compound with formula O8P2Sr3. It is a crystalline substance used in medicine and industry.
Strontium Phosphate
O8P2Sr3
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

William Henry
1774
–
1836
William Henry formulated Henry’s Law, describing how the amount of gas dissolved in a liquid is proportional to its partial pressure
“Chemistry reveals the hidden secrets in the invisible”

Dmitri Ivánovich Mendeleev
1834
–
1907
Dmitri Mendeleev formulated the periodic table of elements, organizing known ones and predicting unknown ones with remarkable accuracy.
“In science, truth is revealed sooner through errors than through confusions”
Become a giant
Your path to becoming a giant of knowledge begins with these top free courses

Free mode

Introduction to Solid State Chemistry


Free mode

Preparing for CLEP Chemistry: Part 1


Free mode

Big Bang and the Origin of Chemical Elements


Free mode

Pre-University Chemistry

Professional development for Educators
Your path to becoming a giant of knowledge begins with these top free courses

Free mode

HP Digital Skills for Educators – Microsoft 365 Copilot


Free mode

How to Learn Online


Free mode

Teaching With Technology and Inquiry: An Open Course For Teachers


Free mode

Support kids’ projects: Programming with Scratch