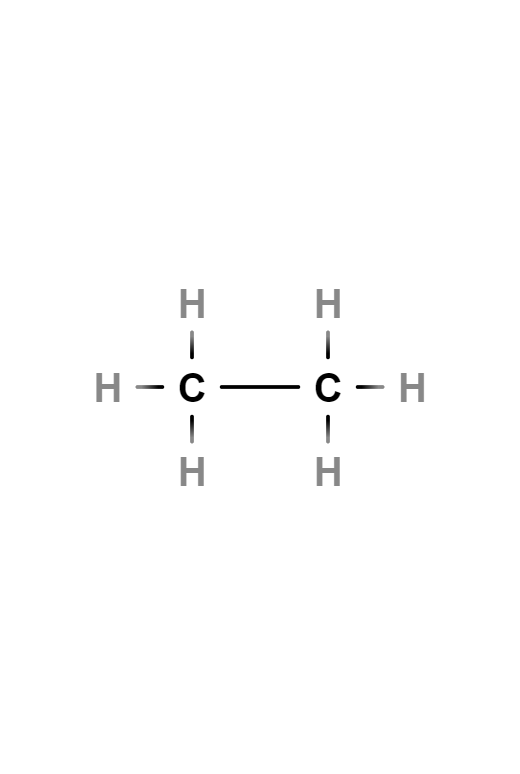Alkane Hydrocarbons. Nomenclature and formulation
The online 3D alkane hydrocarbon molecules of this page visualize through 3D images of the molecules of some of the most important hydrocarbons of this type: methane, ethane, propane and butane. We will discover what are alkane hydrocarbons, how is their nomenclature and formulation, as well as their presence in nature and their applications

This Thematic Unit is part of our Chemistry collection

STEM OnLine mini dictionary
Alkane
Hydrocarbon composed only of carbon and hydrogen atoms joined by single bonds.
Alkyl Group
Structure resulting from removing a hydrogen atom from an alkane, allowing it to attach to a main chain as a branch.
Homologous Series
Group of organic compounds with the same chemical function that differ only by the number of methylene groups.
Isomer
Compounds that have the same molecular formula but different structure or spatial arrangement of their atoms.
Paraffin
Technical name for alkanes highlighting their low reactivity towards most chemical reagents.
Saturated Hydrocarbon
Organic compound containing the maximum possible number of hydrogen atoms per carbon, with no double or triple bonds.
Unsaturated Hydrocarbon
Hydrocarbon containing at least one double or triple bond between its carbon atoms.
What are alkane hydrocarbons
Alkane hydrocarbons are a class of fundamental organic compounds consisting of carbon and hydrogen atoms only. They are known as saturated hydrocarbons because all the bonds between the carbon atoms are single, meaning that they are saturated with hydrogen atoms.
Nomenclature and formulation of alkane hydrocarbons
These compounds are named according to the number of carbon atoms present in their molecule. For example, methane has only one carbon atom, ethane has two carbon atoms, propane has three carbon atoms and so on.
Their chemical structure is based on a linear or branched carbon chain, where hydrogen atoms are linked to carbon atoms by single bonds. The general formula for alkanes is CnH2n+2, where “n” represents the number of carbon atoms.
Presence of alkane hydrocarbons in nature
Alkanes can be found in different states of matter, from gases such as methane to liquids such as pentane and hexane, and even solids such as heptane. Alkane hydrocarbons are widely found in nature, forming a major part of natural gas deposits, where short-chain hydrocarbons such as methane, ethane, propane and butane predominate. Long-chain alkanes are mainly present in petroleum. They are also generated biologically in oxygen-depleted environments, such as swamps or the seabed, where methane is produced by bacteria. They can also be found in small quantities in living organisms, as part of plant waxes and certain oils. This diversity of origins underlines the relevance of alkanes both industrially and ecologically.
Applications of alkane hydrocarbons
Alkanes are widely used as fuels. Natural gas, which contains mainly methane, is an important energy source used for heating, electricity generation and as a fuel in vehicles. Gasoline, which is a mixture of hydrocarbons, also contains alkanes, such as heptane and octane, which provide energy for internal combustion engines.
In addition to their use as fuels, alkanes have applications in the petrochemical industry. They are used as feedstock for the production of plastics, such as polyethylene and polypropylene, as well as for the manufacture of chemicals, lubricants, solvents and detergents.
In summary, these online 3D alkane hydrocarbon molecules give you a visualization of the molecules that will undoubtedly help you to better understand these important chemical compounds. Don’t miss them!

STEM OnLine mini dictionary
Alkane
Hydrocarbon composed only of carbon and hydrogen atoms joined by single bonds.
Alkyl Group
Structure resulting from removing a hydrogen atom from an alkane, allowing it to attach to a main chain as a branch.
Homologous Series
Group of organic compounds with the same chemical function that differ only by the number of methylene groups.
Isomer
Compounds that have the same molecular formula but different structure or spatial arrangement of their atoms.
Paraffin
Technical name for alkanes highlighting their low reactivity towards most chemical reagents.
Saturated Hydrocarbon
Organic compound containing the maximum possible number of hydrogen atoms per carbon, with no double or triple bonds.
Unsaturated Hydrocarbon
Hydrocarbon containing at least one double or triple bond between its carbon atoms.
Explore the exciting STEM world with our free, online, simulations and accompanying companion courses! With them you’ll be able to experience and learn hands-on. Take this opportunity to immerse yourself in virtual experiences while advancing your education – awaken your scientific curiosity and discover all that the STEM world has to offer!
Methane
Methane is the simplest alkane hydrocarbon, whose chemical formula is CH4. Each of the hydrogen atoms is bonded to carbon by a covalent bond. It is a non-polar substance that occurs as a gas at ordinary temperatures and pressures. It is colorless, odorless and insoluble in water.
Methane
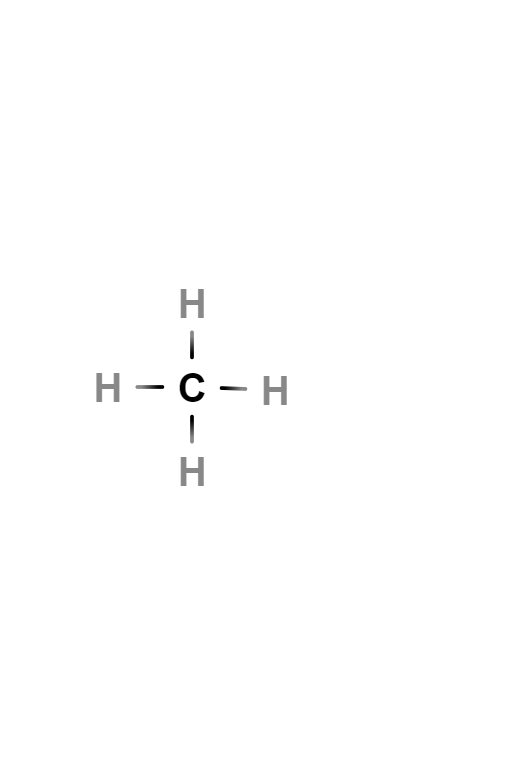
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
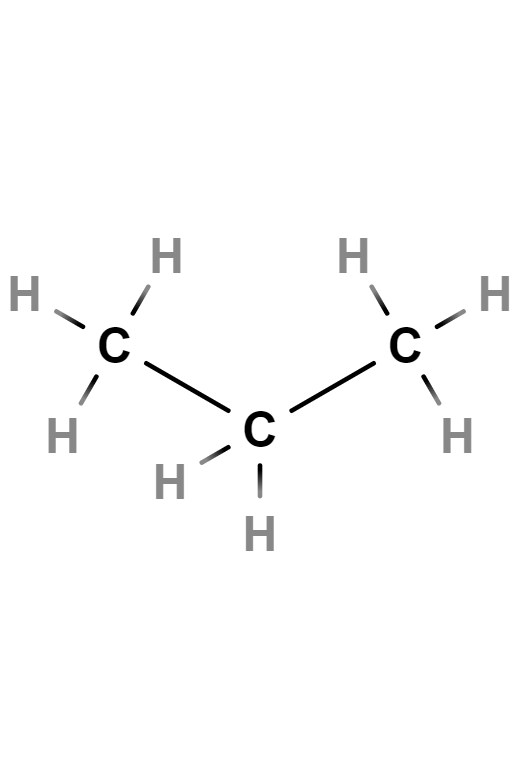
Butane
Butane, also called n-butane, is a saturated, paraffinic or aliphatic, flammable, gaseous hydrocarbon that liquefies at atmospheric pressure at -0.5 °C, consisting of four carbon atoms and ten hydrogen atoms, whose chemical formula is C4H10. An isomer of this gas can also be called by the same name: isobutane or methylpropane.
Butane
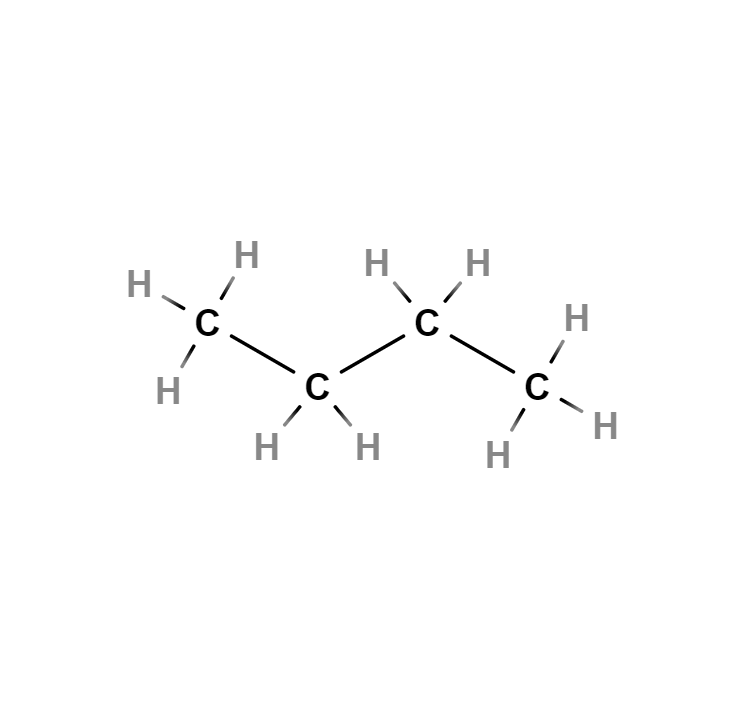
 Single bond
Single bond
Double bond
 Triple bond
Triple bond
Wedge bond

Hash bond
Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Friedrich Wöhler
–
Become a giant


Principles of Biochemistry



Medicinal Chemistry: The Molecular Basis of Drug Discovery



Introduction to Pharmacology



Biorefinery: From Biomass to Building Blocks of Biobased Products



Big Bang and the Origin of Chemical Elements



Pre-University Chemistry



Preparing for CLEP Chemistry: Part 1

Professional development for Educators


Introduction to Data Wise: A Collaborative Process to Improve Learning & Teaching



Teaching with Physical Computing: Soft skills, teamwork and the wider curriculum



Learning How to Learn: Unlocking a Growth Mindset with AI



HP AI Teacher Academy

Giants of science
“If I have seen further, it is by standing on the shoulders of giants”
Isaac Newton

Friedrich Wöhler
–
Become a giant


Principles of Biochemistry



Medicinal Chemistry: The Molecular Basis of Drug Discovery



Introduction to Pharmacology



Biorefinery: From Biomass to Building Blocks of Biobased Products



Preparing for CLEP Chemistry: Part 1



Big Bang and the Origin of Chemical Elements



Pre-University Chemistry

Professional development for Educators


Teaching with Physical Computing: Soft skills, teamwork and the wider curriculum



Teaching With Technology and Inquiry: An Open Course For Teachers



HP AI Teacher Academy



Teach computing: Physical computing with Raspberry Pi and Python

Test your knowledge
What are alkane hydrocarbons, and how are they chemically structured?
How are alkane hydrocarbons named, and what is their general formula?
Where are alkanes found in nature, and why are they important?
What are alkane hydrocarbons used for?
What characteristics make alkanes useful in industry?
You may also be interested
Last simulations
To learn and experience

Take your knowledge to the next level with science kits and hands-on tools that connect theory with experimentation

Periodic table

Teachers essentials

Notion
Organize your notes, tasks, and projects in one place. Notion combines notes, lists, and calendars in one flexible space.

Canva
Design presentations, diagrams, and infographics with easy-to-use templates and professional results

Genially
Create interactive content, presentations, and educational games in a simple and visual way

Desmos
The most intuitive graphing calculator to explore mathematics and create dynamic classroom activities

Tinkercad
Design in 3D and simulate electronic circuits for free; it’s the perfect tool for simple robotics projects

Your Infinite Library
Enjoy the freedom to explore millions of titles and a curated selection of magazines on any device


Join Prime for students and young adults
The best series. Millions of songs. All included with your subscription.

Science made fun
Learn with the best courses

edX
Online courses from leading universities and companies. Learn practical skills and earn professional certifications

Coursera
Courses from leading universities and companies. Learn practical skills and earn professional certifications

Udemy
Thousands of online courses tailored to your level. Learn at your own pace with expert instructors